
Here’s a diagram I made of how ATP is produced in glucose metabolism: It produces NADH, FADH 2, and GTP, which enter the electron transport chain to produce ATP. The CAC’s contribution to cellular ATP supplies is indirect. 
This activity is higher in the liver and kidney than elsewhere. Under these conditions, the CAC is not replenishing its own supply of OAA, and so pyruvate carboxylase activity is key. In this process, intermediates from other processes enter the CAC and then oxaloacetate is siphoned off to create glucose. That process is especially important in the liver and kidney because they perform gluconeogenesis. Under this circumstance, acetyl-CoA feeds back to activate pyruvate decarboxylase, thus causing more OAA to be generated and more CAC cycles to run in parallel. This is especially important when intermediates are being rapidly siphoned off for other processes, causing acetyl-CoA to accumulate while very few of the CAC cycles are actually running to completion. Converting pyruvate to oxaloacetate and thus increasing the number of parallel CAC jobs is a way of increasing the rate at which intermediates are produced for other cellular processes. In other words, while acetyl-CoA is just consumed in the CAC, oxaloacetate is consumed and then regenerated (the cycle starts and ends with it), and so the amount of oxaloacetate available can be limiting and thus determine the number of instances of the citric acid cycle that can run in parallel. Pyruvate can also be converted to oxaloacetate by pyruvate carboxylase- think of that as adding a node to the computing cluster. Think of that as submitting a job to the computing cluster. As discussed above, it can be spent on creating acetyl-CoA. 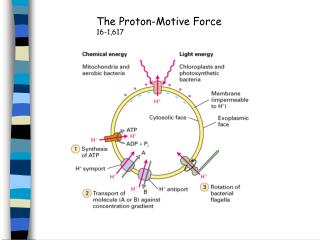
Pyruvate can actually feed into the cycle in two ways. The diagram at 0:06, which should be the thumbnail below, is a concise overview. Here’s a video describing these roads in and out of the CAC. The CAC is described as “amphibolic” because it is both catabolic and anabolic – it both produces and consumes intermediates relevant to a host of other pathways. Citrate, an intermediate in the CAC, inhibits PFK-1, a step in glycolysis. It also makes sense to have glycolysis and CAC running at about the same rate (think just in time delivery), so there’s a mechanism for their co-regulation. for instance, succinyl-CoA, an intermediate, competes with acteyl-CoA for the attention of citrate synthase. for instance ADP (whose accumulation is a sign of energy need) activates enzymes such as the pyruvate dehydrogenase complex (above table) for instance NADH not only inhibits the pyruvate dehydrogenase complex (above table) but also inhibits other regulatory steps. 
availability of oxaloacetate and acetyl-CoA as inputs. Orthogonally, you can think of 4 main mechanisms by which the CAC is regulated: Three other highly exergonic CAC steps are also checkpoints for regulation: citrate synthase, isocitrate dehydrogenase, and the alpha ketoglutarate dehydrogenase complex. the / ratio and / ratios are high, there’s no need to continue running the cycle. Tl dr: when ample fuel is available, i.e. It is tightly regulated by allosteric regulation by products of the CAC / electron transport chain: Amino acid catabolism or fatty acid catabolism can also yield acetyl-CoA directly.Īs the pyruvate dehydrogenase complex is the chief entry point into the CAC, its proper regulation is key for controlling the rate of cellular energy production. Pyruvate can come either from glycolysis or amino acid catabolism. What’s not depicted in this diagram is there are a few ways to enter the CAC. Source: this is a Wikimedia Commons featured image by multiple authors. Pyruvate dehydrogenase deficiency causes a neurodevelopmental disorder and can result from mutations in many of the different genes involved in the complex. This can be considered as step 0 and is depicted at the top of the below diagram. First it passes through a “transition phase” where it is undergoes oxidative decarboxylation to CO 2 at the hands of the pyruvate dehydrogenase complex, transferring acetyl groups to coenzyme A to yield acetyl-CoA. Pyruvate from glycolysis does not enter directly into the citric acid cycle. It takes place inside the mitochondrial matrix. It’s called a “cycle” and not a “pathway” because it both begins and ends with oxaloacetate. The citric acid cycle or Kreb’s cycle is the cell’s metabolic hub, as it not only generate energy from pyruvate (the product of glycolysis) but along the way also can consume and produce metabolites relevant to host of other processes. Here’s a rad CC BY Wikimedia Commons diagram by Kelvinsong & Sowlos: To understand this post, it’s useful to know the mitochondrial structure ( see also Cell Biology 03). These are notes from lecture 8 of Harvard Extension’s biochemistry class. Biochemistry 08: the citric acid cycle and the electron transport chain
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
